
Award to our Anne Berends for best Scientific Publication at LpS Digital
Life Science / Sunled
Life Science / Sunled / January 23, 2022

Photobiomodulation or PBM is the term to describe light-induced reactions in our body. Specifically, molecules in our skin cells absorb certain wavelengths of light which results in activation of biochemical processes. Thus, although PBM is induced by absorption of light, our eyes do not play a role in this process.
The history of using light as form of treatment for disease or ‘photomedicine’ is claimed to go back as far as 1400 BC.1 In those days and in the 1700’s, 1800’s and early 1900’s, photomedicine to treat all kinds of skin diseases and wounds was mostly making use of sunlight.1 In the second half of the 20th century, after the discovery of lasers, these devices were started to being used in medicine, and it was merely a coincidence that the promotion of wound healing as an effect of low intensity ruby laser irradiation was found by Prof. Mester in 1965.1
From this first observation many follow up experiments and publications followed, showing the beneficial effect of low intensity red laser light on healing of skin wounds, regeneration of muscle fibers, and treatment of skin necrosis:1 the field of low level laser (light) therapy (LLLT) was born. Over the years, many more scientific studies were published (Figure 1) and a wide variety of treatment devices for professional and home use entered the market. More recently the scientific community adopted the term photobiomodulation (therapy) or PBM(T), to describe the effect of light on cells, as light emitting diodes (LEDs) are just as good as lasers for this application and the word ‘low’ is regarded as too arbitrary.2
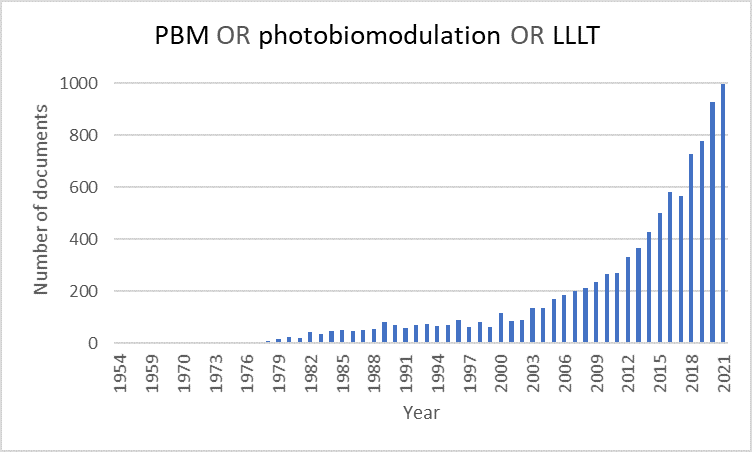
Figure 1. Scopus data, December 2021.
In the scientific community, a wide variety of beneficial effects of red or near-infrared light on the human body is attributed to PBM.3,4 Examples include treatment of retinal diseases,5 oral muscocitis,6 wound healing,7 bone repair,8,9 pain relief,10 brain treatment like Parkinson’s disease, stroke, brain trauma, and depression,11 sleep quality,12 muscle performance,13 sports recovery14, an hypothesis for COVID-19 treatment,15 and probably many more.
Although many reports on the beneficial effects of PBM exist, the biological mechanisms that result in this wide variety of effects is not yet completely understood. Many scientists agree that the PBM process starts with red and near-infrared light that activates the mitochondria, the part of our cells that produces energy.15–19 Most of them refer to cytochrome c oxidase specifically, a protein complex that is part of the electron transport chain in the mitochondria that finally results in energy production (or ATP)15–17,20,21 while others refer to (nano)confined water molecules inside the membranes of mitochondria that are affected by red and near-infrared light.18,19,22,23 Side products of the energy production process in mitochondria are nitric oxide (NO) and reactive oxygen species (ROS). These molecules in turn serve as signaling molecules in the body, activating hundreds of other cascade processes.
Another factor that makes a well-structured discussion on biological mechanisms and effects of PBM complicated, is the wide variety of treatment parameters that is used in scientific studies. Not only different wavelengths or combinations thereof are studied (usually between 600 and 1100 nm), studies also differ in intensity (i.e. average and peak irradiance), pulsing or continuous wave, pulse characteristics (frequency, duty cycle, pulse width), irradiated area (cm2), duration, radiant energy (J), dose (J/cm2), in vivo or vitro, in healthy young adults or people with health problems, etc.23,24 This makes it difficult to come to a systematic analysis of factors that play a major or minor role in the PBM mechanism or to determine which factors are important to induce a specific effect.
It is thus clear that although many beneficial health effects are linked to red and near-infrared light, the exact mechanism and treatment parameters are still under investigation. This is why Seaborough decided to hire an independent third party to organize a clinical trial to study the treatment parameters of near-infrared radiation on the health and well-being effects on generally healthy people.25 In this way, we could determine the minimum dose requirement to achieve a measurable and significant effect on our participants. The results of this clinical trial, showing significant effects on resting heart rate and immune parameters, will be published in a peer-reviewed scientific journal, expected in 2022. In the meantime, we developed the SunLED technology further so it can be easily implemented in modern lighting solutions.